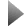10-05/06 :: May/June 2010
nanotimes Patents
Image left: Common structural formula of the monoetherified diols of the diamondoids according to the present invention in which − D represents a diamondoid, − -CHR1R2 represents a protective group in which R1 and R2 are linear or branched alkyl groups and wherein the protective group contains at least one halogen atom,
and R3, R4, R5 and R6 represent independent of one another:
− hydrogen, − a linear or branched alkyl group with 1 to 20 carbon atoms,
− a cyclic alkyl group with 3 to 20 carbon atoms, − an aryl group with 6 to 18 carbon atoms − a heteroaryl group with 5 to 18 ring atoms in which 1 or 2 atoms are heteroatoms, chosen from nitrogen, oxygen, sulphur, and the remaining ring atoms are carbon atoms.
Contact: TransMIT GmbH, Dr. Kerstin Lischka, Kerkrader Str. 3, D-35394 Giessen, Germany, Phone: +49-(0)641- 94364-25: http://www.transmit.de
http://www.hipo-online.de/files/Synopsis_Diamando- id_Diols_TM285_EN_070410.pdf
73
Ortho-metalated, chelate-stabilized ben- zylamines of the rare-earth metals (RE)
Inventors: Prof. Dr. Jörg Sundermeyer, Dr. Alexander Petrov, Oliver Thomas
Abstract: The invention at hand provides, for the first time, thermally stable trisaryl compounds of the rare earth metals which are storable for a long time. These complexes may be produced in situ from the corresponding ortho-lithiated aryl ligands and an anhydrous rare earth metal halide at room tempe- rature and under an inert atmosphere. Benzylamine complexes of the rare earth metals according to the present invention may be used as catalysts for the hydroamination of olefins.
In the context of the present invention, rare earth metals comprise not only 4f elements, but also scan- dium (Sc) and yttrium (Y).
Advantages and special characteristics • The ortho-metalated homoleptic benzylamine complexes of the rare-earth metals according to the present invention can be produced in situ by reaction of an ortho-lithiated aryl ligand with a rare-earth metal halide;
• The method is suitable even for large cations, e.g. Sm3+ and Gd3+;
• The complexes are thermally stable and storable for a long time;
• Suitability as catalysts for the hydroamination of olefins;
• Significantly enhanced catalytical activity as
 Previous Page
Previous Page